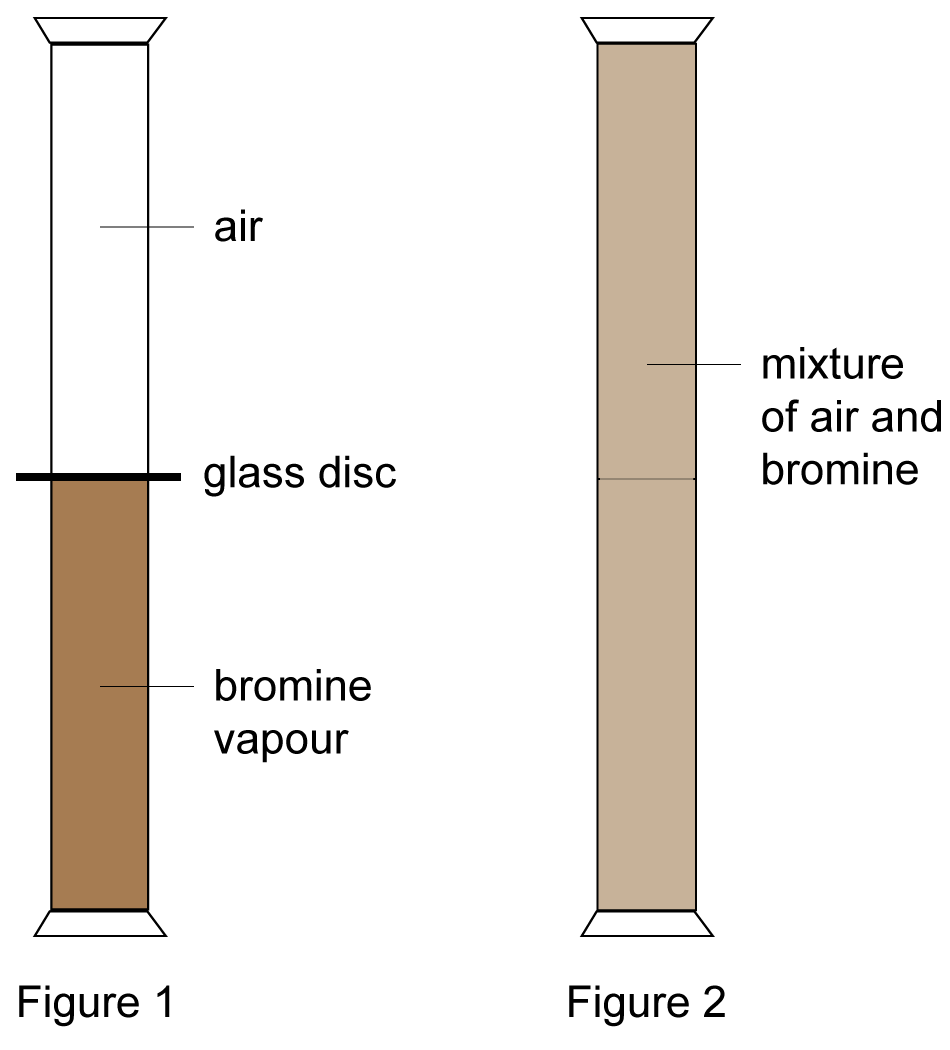IGCSE Chemistry 2017: 1.3: Understand How the Results of Experiments Involving the Dilution of Coloured Solutions and Diffusion of Gases can be Explained

GASES LIQUIDS SOLIDS States of Matter, kinetic particle theory models diagrams state changes, melting, boiling, evaporation, condensing, freezing, solidifying, cooling curves particles pictures elements compounds mixtures heat conduction electrical ...

Bromine gas diffusion. Bromine vapour (orange) filling a gas jar as it evaporates from bromine liquid (dark brown, bottom of jar). Because gas molecul Stock Photo - Alamy

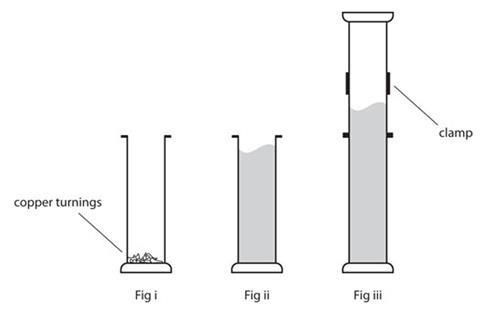
Bromine vapour is heavier than air Even soit spreads upwards in the following experiment Why Place an open gas jar of air upside down on an open gas jar containing a few

A bottle of perfume is opened in the room why can we smell it after a while This is called diffusion Diffusion is when particles move from a high. - ppt download

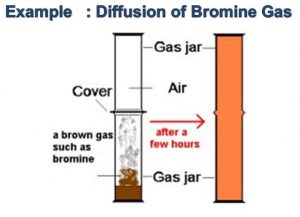
When a gas jar full of air is placed upside down on a gas jar full of bromine vapours, the red - brown vapours of bromine from the lower jar go upward