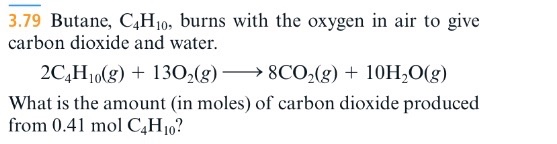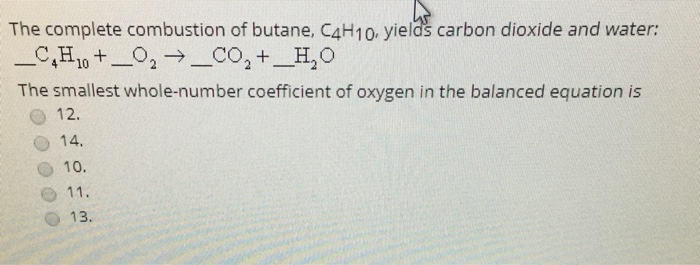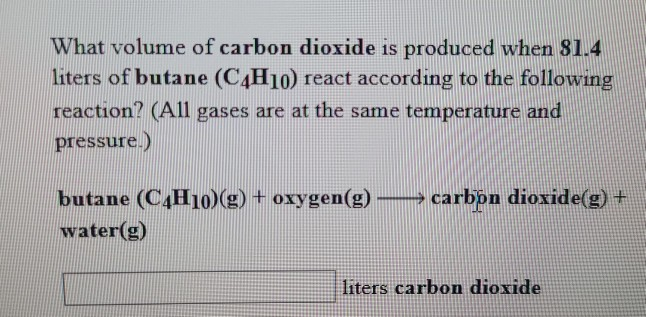
SOLVED: Gaseous butane, C4H10, reacts with diatomic oxygen gas to yield gaseouscarbon dioxide and water vapor.

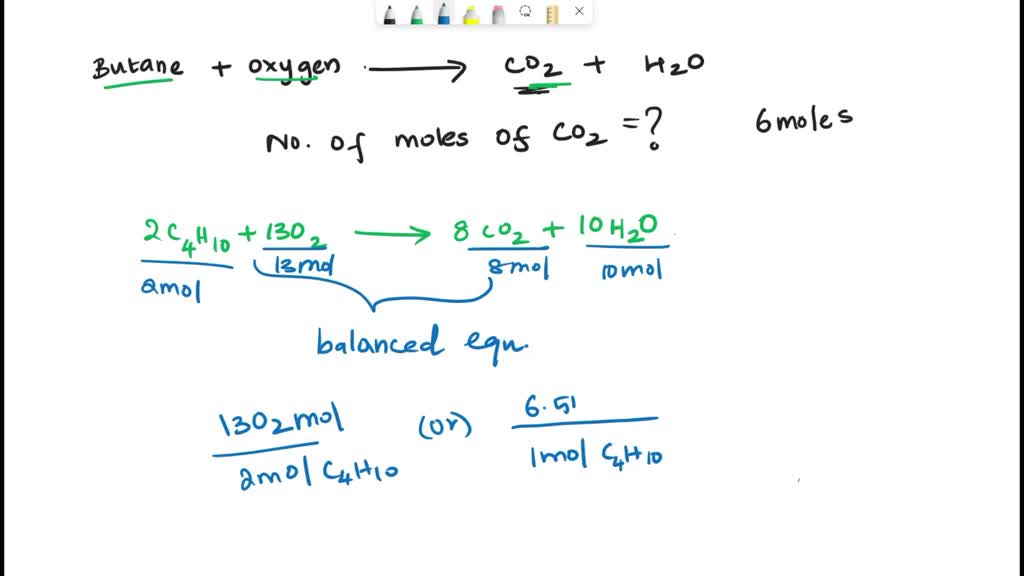
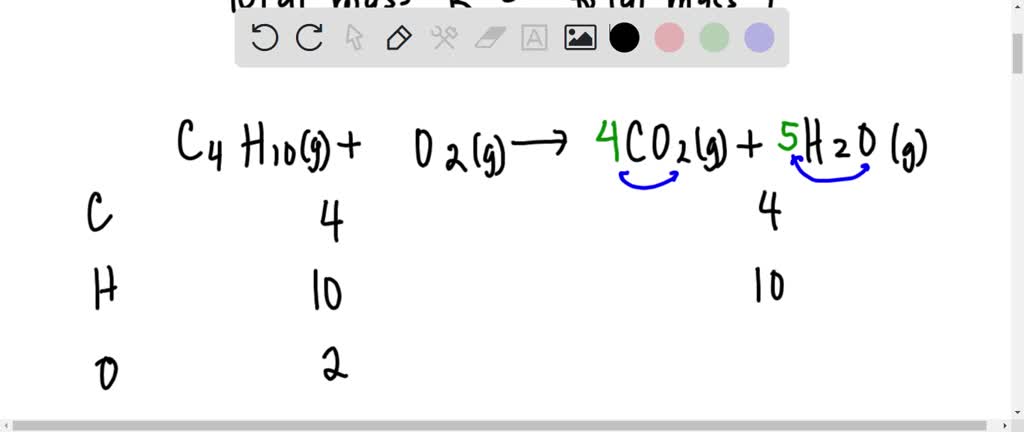
SOLVED: The balanced chemical equation for the reaction between butane (C4H1o) and oxygen is: 2 C4H1o ( g ) + 13 02 ( g ) 8C02 ( g +10 Hz0 ( g )

Butane (C(4)H(10)) gas burns in oxygen to give carbon dioxide and water accoding to the reaction. 2C(4)H(10) (g) + 13O(2)(g) rarr 8 CO(2)(g) + 10H(2)O(l) When 5.0 L of butane ware burnt

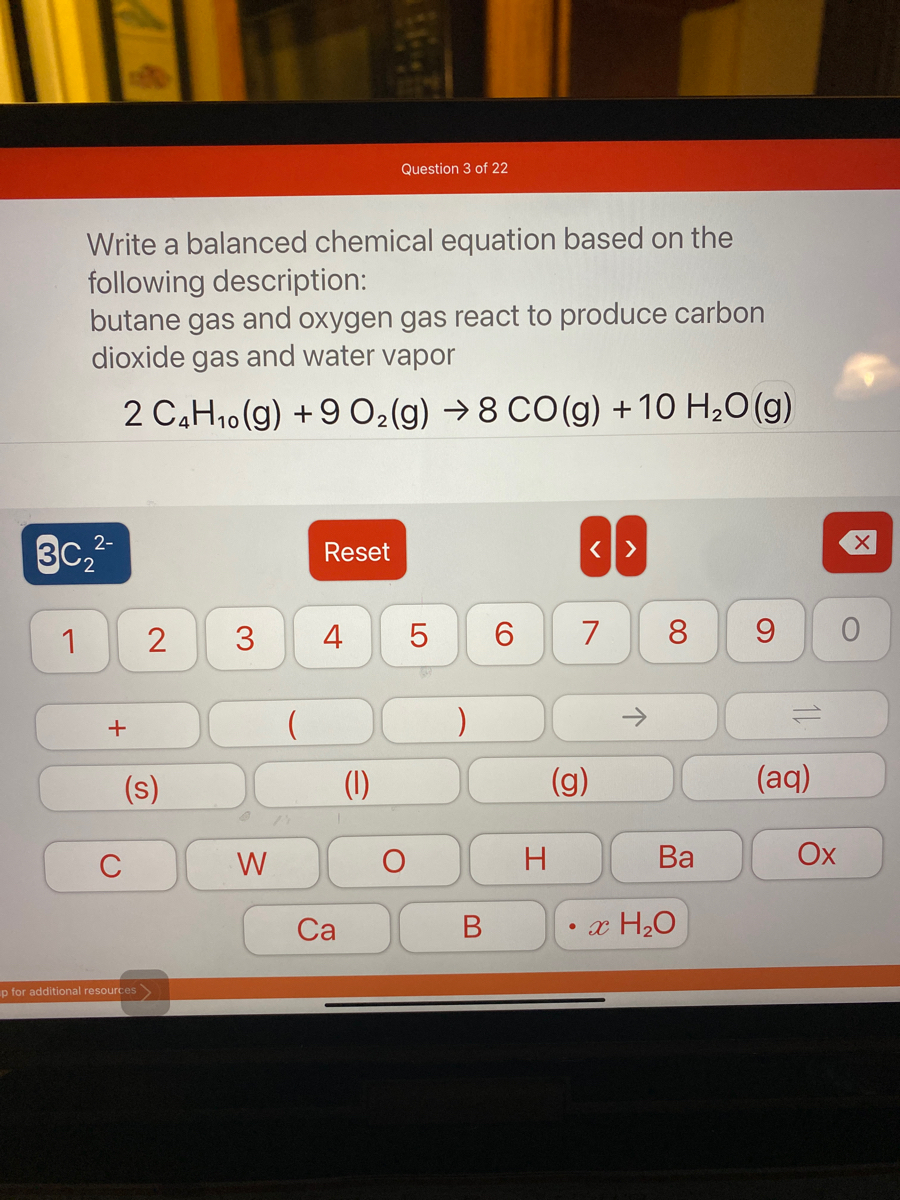
SOLVED: Now balance the chemical reaction by providing the correct whole number coefficients: butane (C4H1o) + oxygen gas yields carbon dioxide water ? JC4H1o JOz 7 Jcoz + HzO Enter

When 0.340 mol of butane, C_4H_{10}, are burned with excess oxygen giving CO_2 and H_2O, how many moles of oxygen are consumed? | Homework.Study.com

When butane burns in oxygen, it produces carbon dioxide and water. This reaction is represented in - Brainly.com