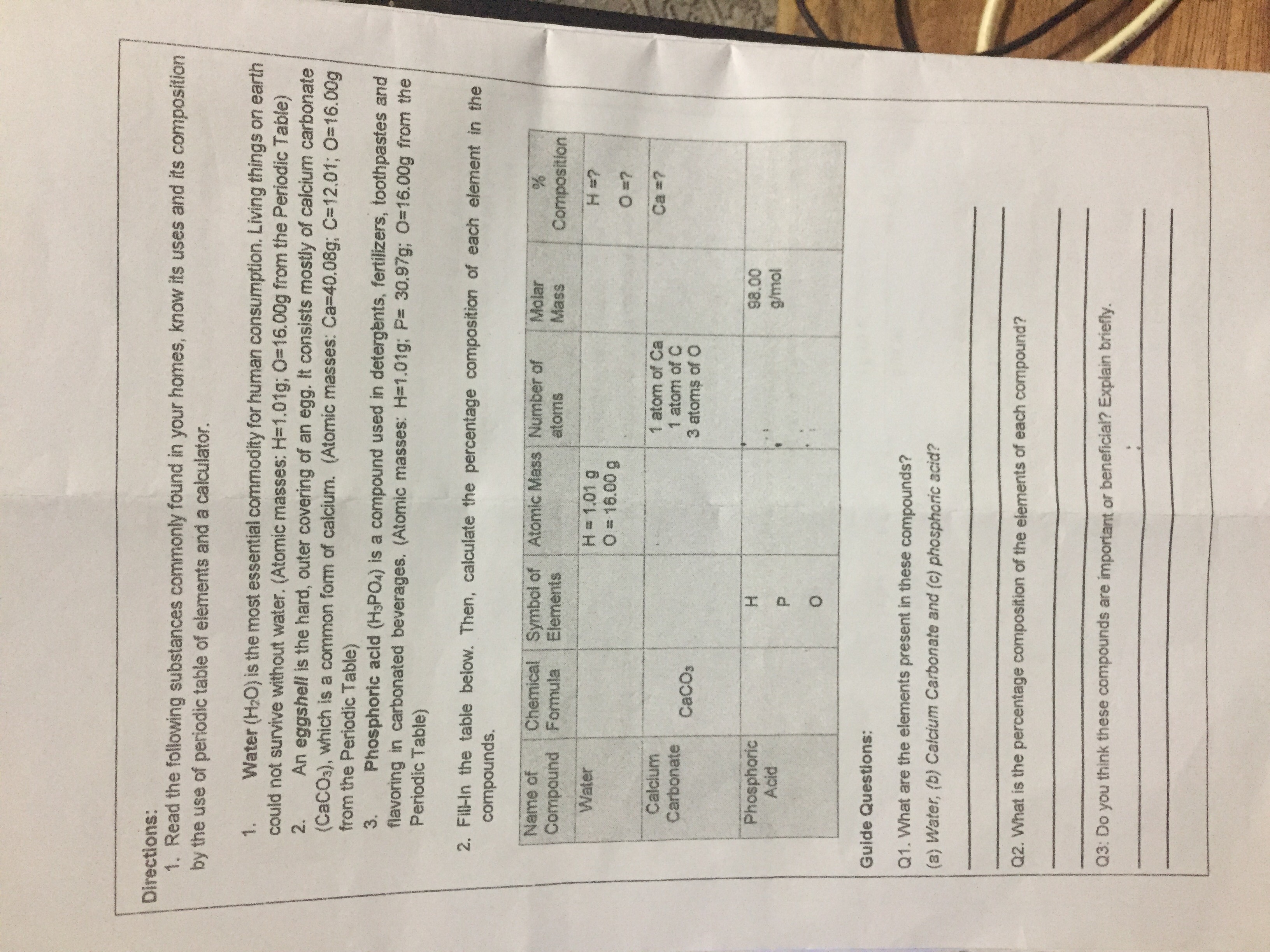
Calculate the % of each element in calcium carbonate. (Atomic mass: C – 12, O – 16, Ca – 40) - Sarthaks eConnect | Largest Online Education Community
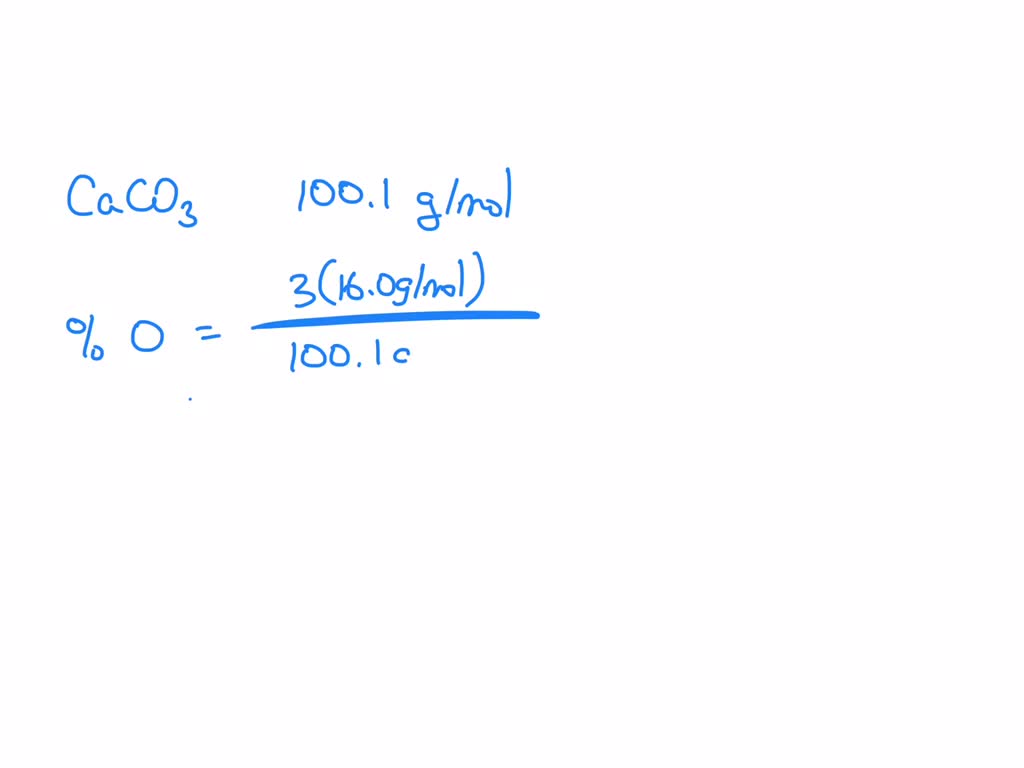
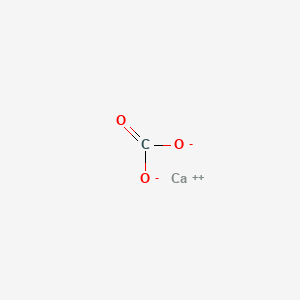
SOLVED: The formula of calcium carbonate Is CaCO3 Determine the percent composition of oxygen in this compound: Round answer t0 three significant figures: Provide your answer below:
20 g of an impure sample of calcium carbonate decomposes on heating to give 8.4 g of calcium oxide.What is percentage purity of calcium carbonate sample(at.wt.of Ca=40) a)60 b)75 c)85 d)95

Calculate the mass per cent of each element present in the molecule of calcium carbonate - CBSE Class 9 Science - Learn CBSE Forum

The percentage of three elements calcium, carbon and oxygen in a sample of calcium carbonate is given as: Calcium = 40%; Carbon = 12.0%; Oxygen = 48% If the law of constant
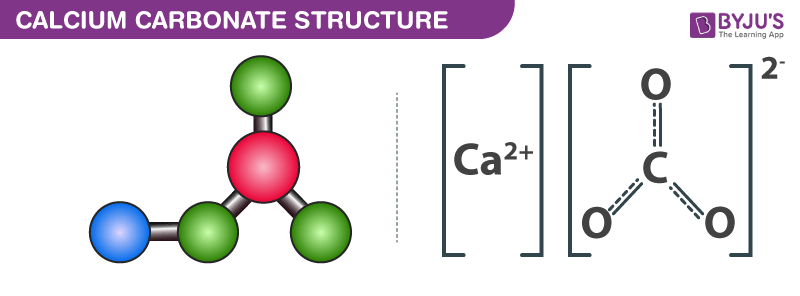
Limestone: Calcium Carbonate (CaCO3) - Uses, Preparation, Properties, Formula & Structure of Calcium Carbonate

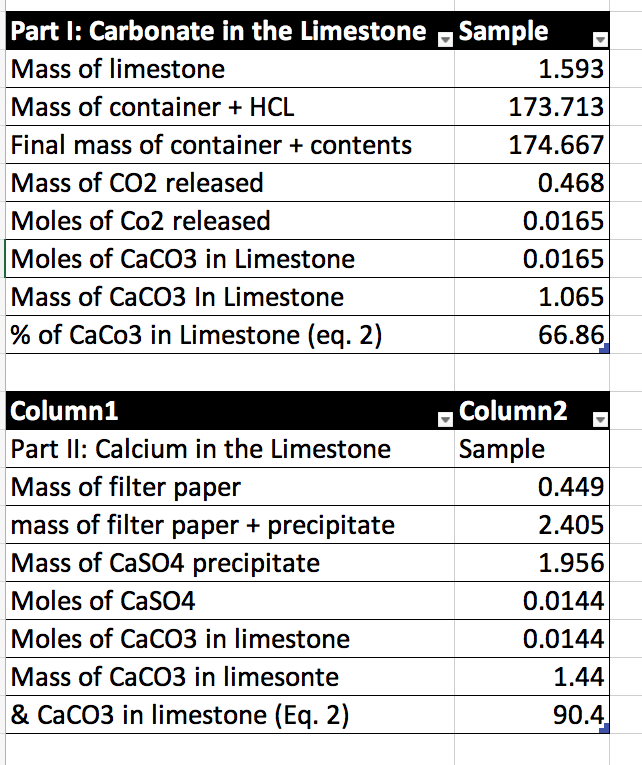






![Telugu] A sample of calcium carbonate (CaCO(3)) has the following per Telugu] A sample of calcium carbonate (CaCO(3)) has the following per](https://static.doubtnut.com/ss/web/7259981.webp)