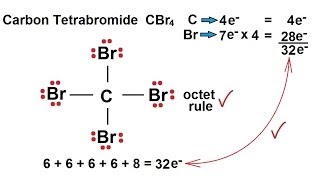
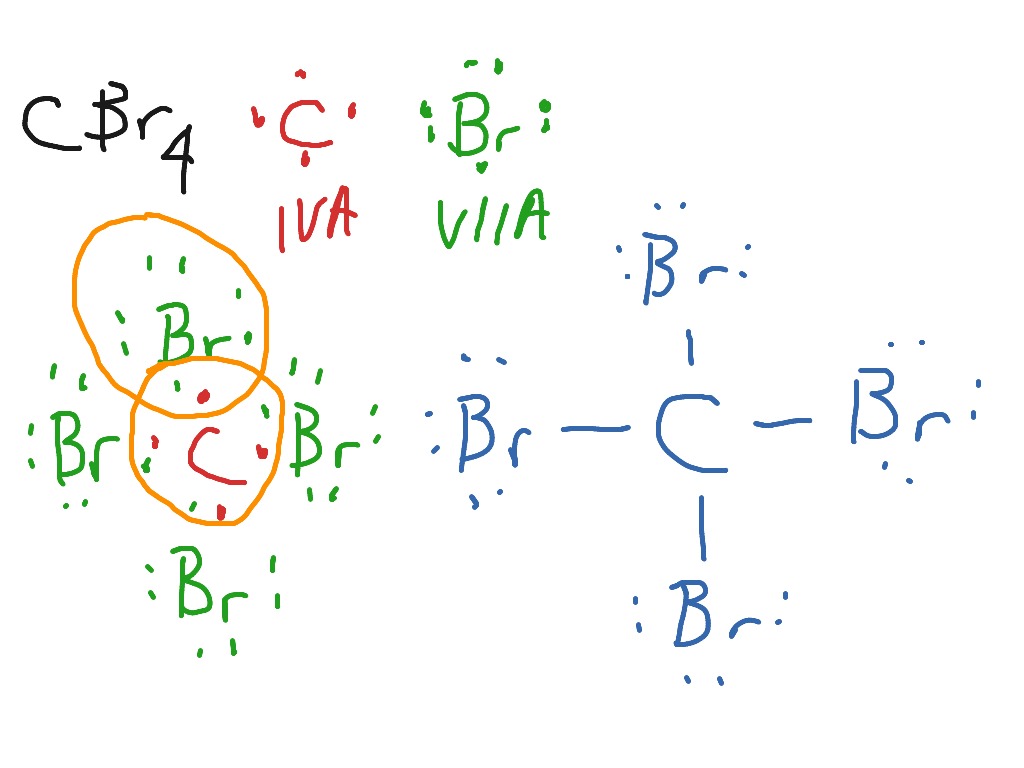A carbon compound contains 12.8% Carbon, 2.1% Hydrogen, 85.1% Bromine. The molecular weight of the compound is 187.9. Calculate the molecular formula.

electrolysis of molten lead bromide products electrode equations anode cathode apparatus electrolyte gcse chemistry KS4 science igcse O level revision notes
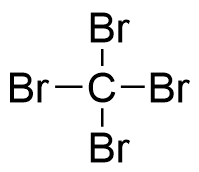
558-13-4 | Carbon Tetrabromide | Carbon Bromide; Methane Tetrabromide; NSC 6179; Tetrabromomethane | CBr₄ | TRC

Equation for the reaction when compound A is bubbled through bromine dissolved in carbon tetrachloride is as follows : A Br2/CCl4 CH2Br | | | CH2Br (i) Draw the structure of A .(

There are four alkyl bromides with the formula C4H9Br. Write their structural formulas and classify each as primary, secondary, or tertiary alkyl bromides. | Homework.Study.com

Cetrimonium bromide antiseptic surfactant molecule. Stylized skeletal formula (chemical structure): Atoms are shown as color-coded circles: hydrogen (hidden), carbon (grey), nitrogen (blue), bromine (brown Stock Photo - Alamy