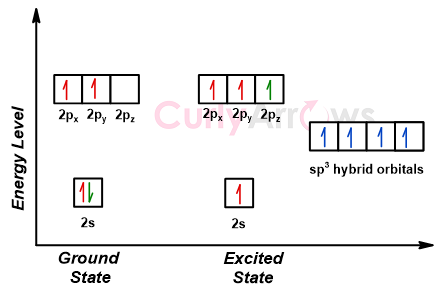
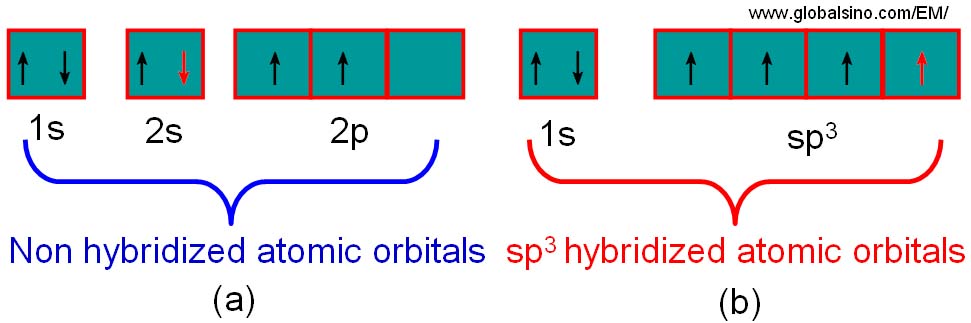
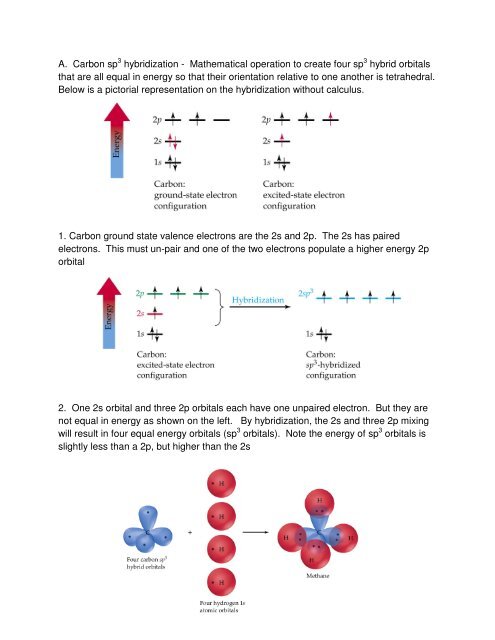
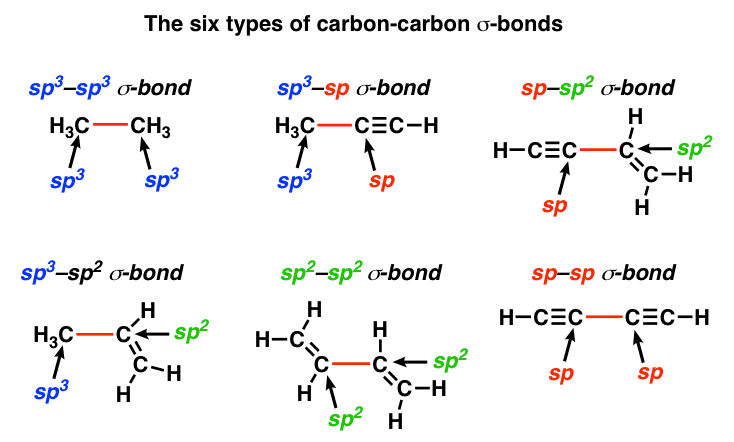
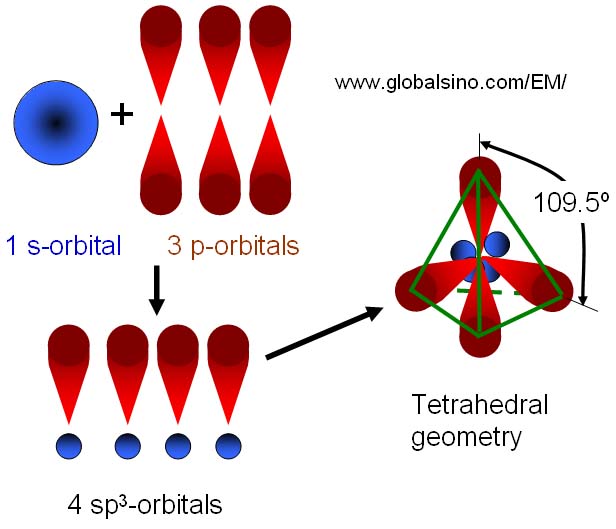
why is this carbon sp3 hybridised? after the reaction, isn't it still sp3, as it has 3 bonding pairs and 1 lone pair? : r/chemistry

Monitoring the thermally induced transition from sp3-hybridized into sp2-hybridized carbons - ScienceDirect
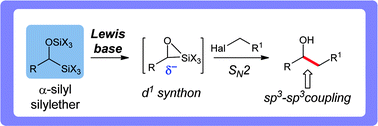
Lewis base-promoted carbon–carbon sp3–sp3 coupling reactions of α-silyl silylethers - Chemical Science (RSC Publishing)

Synthesis and Characterization of Highly Fluorinated Graphite Containing sp2 and sp3 Carbon | Chemistry of Materials

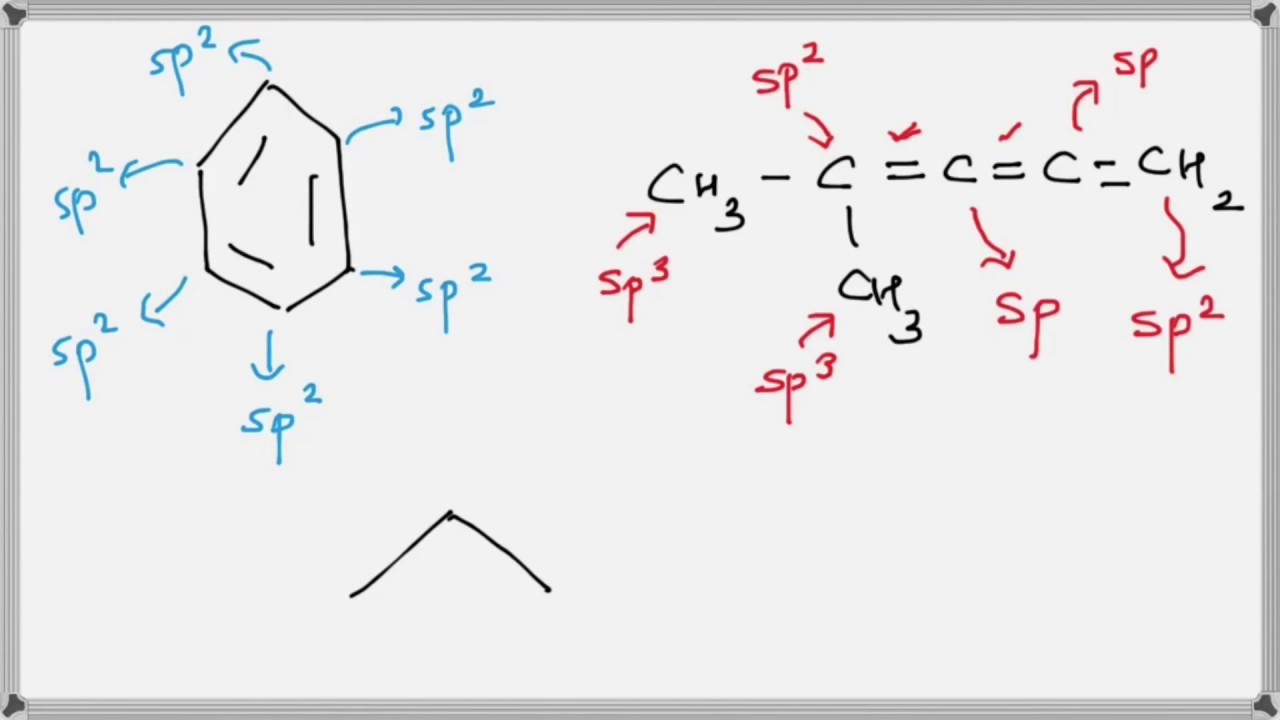
Which of the following represents the given sequence of hybridisation of carbon atoms from left to right : sp^2,sp^2,sp,sp ?

A structure for a molecule that meet the following description: Contains two sp2-hybridized carbons and two sp3-hybridized carbons | Homework.Study.com



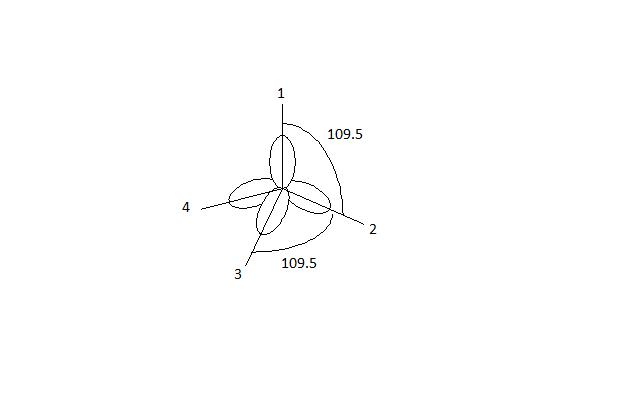




/chapter2/pages1and2/page1and2_files/sp2hyb.png)