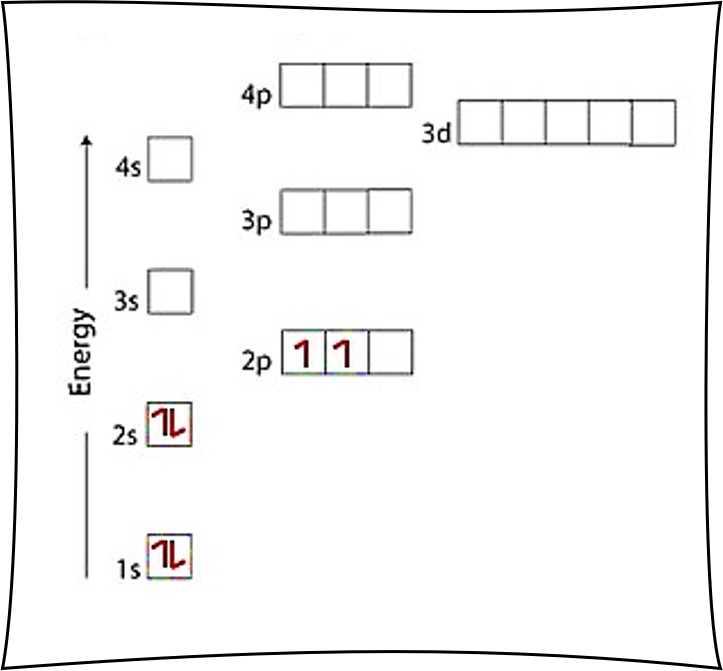
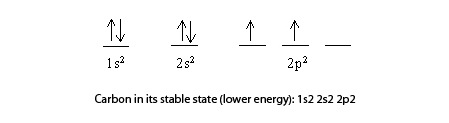
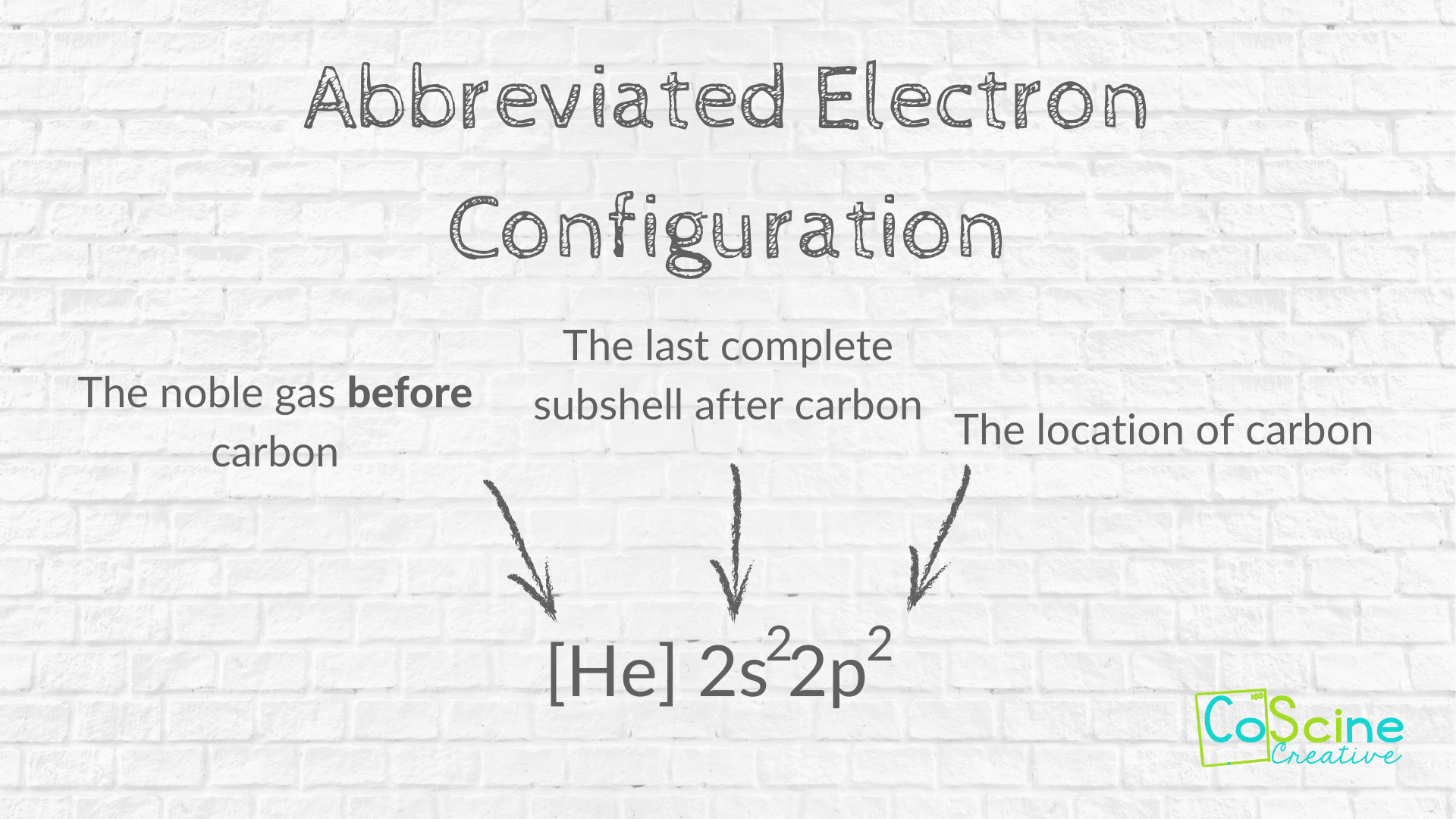
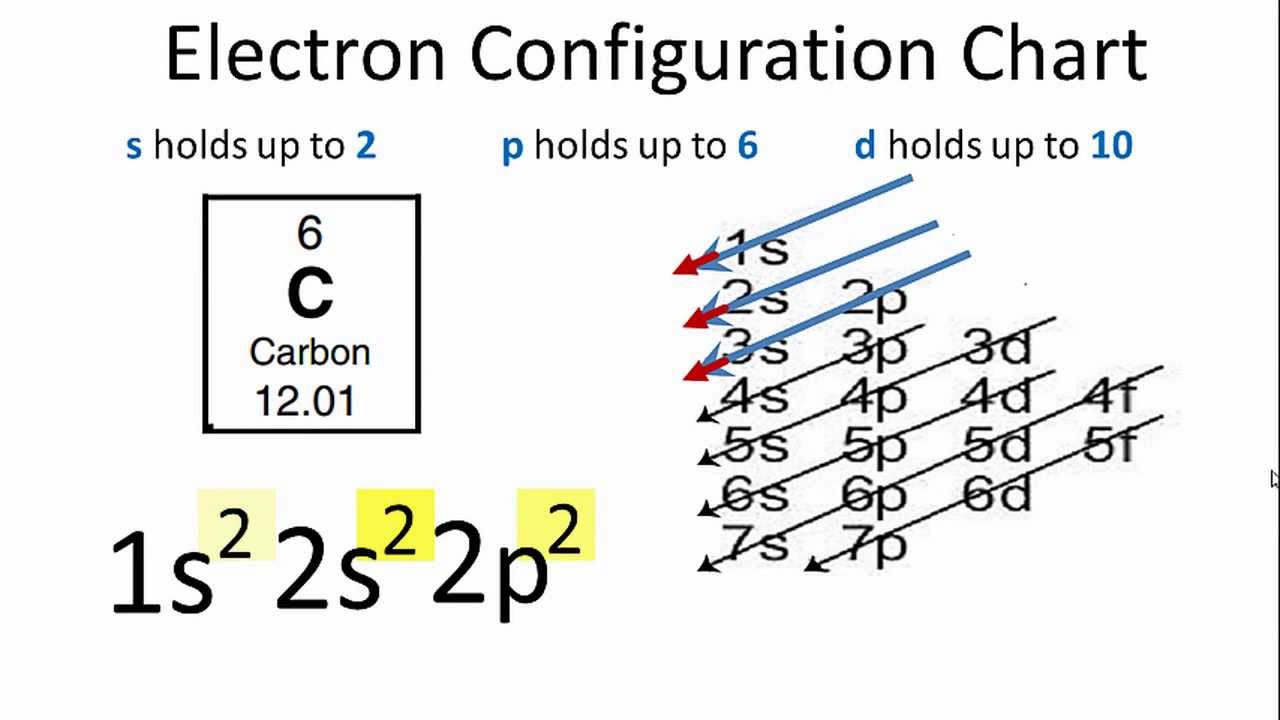
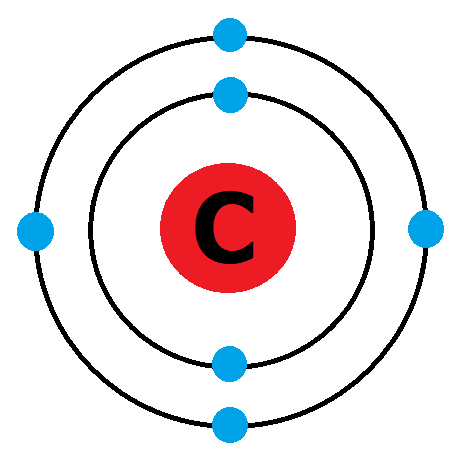
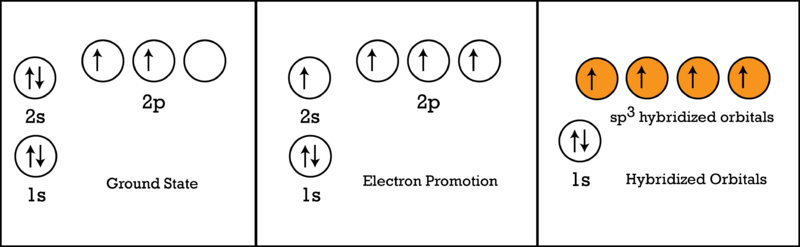
The electronic configuration of carbon atom in the excited state is 1s^{2} 2s^{1} 2p^{3}. Which of the following is/are incorrect statement(s) about it?

Electronic configuration diagram vs energy for carbon atom in its (a)... | Download Scientific Diagram
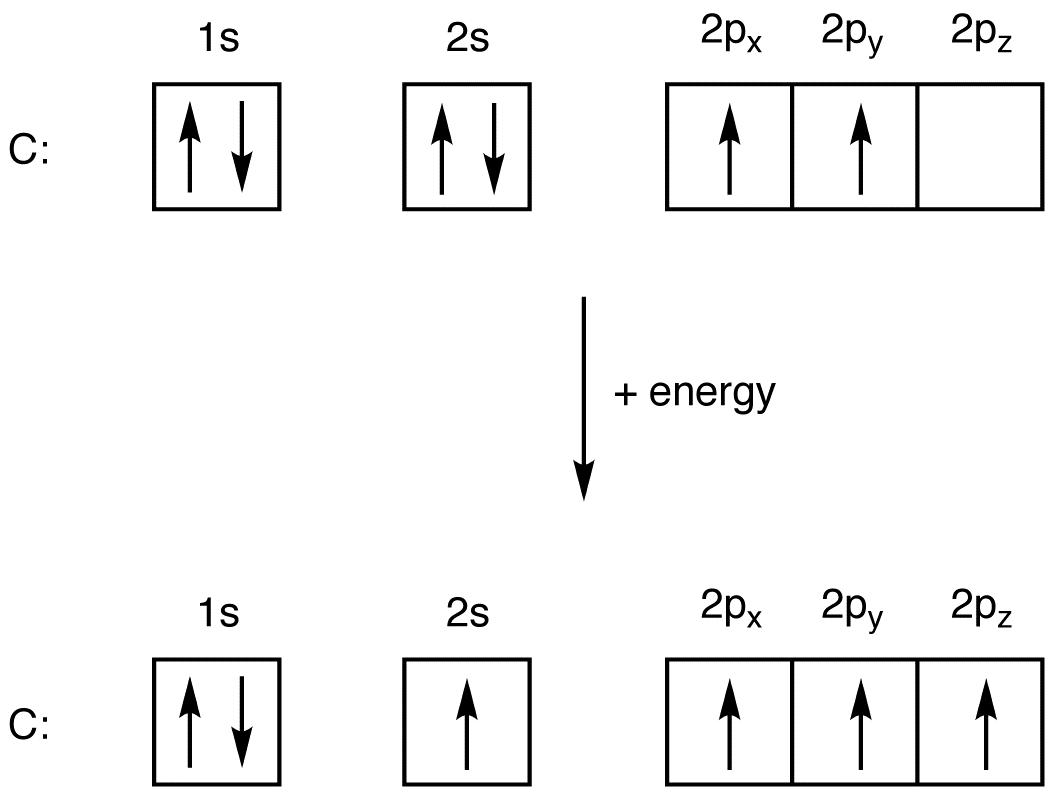
Write the ground state electron configuration for a neutral carbon atom, and for an excited state of carbon? | Socratic
![The electron configuration for the carbon atom is: (a) [He] 2s^22p^2 (b) [He] 2s^4 (c) [Ne] 2s^22p^2 (d) 1s^22p^4 (e) None of these | Homework.Study.com The electron configuration for the carbon atom is: (a) [He] 2s^22p^2 (b) [He] 2s^4 (c) [Ne] 2s^22p^2 (d) 1s^22p^4 (e) None of these | Homework.Study.com](https://homework.study.com/cimages/multimages/16/aufbau7621802225308557580.png)
The electron configuration for the carbon atom is: (a) [He] 2s^22p^2 (b) [He] 2s^4 (c) [Ne] 2s^22p^2 (d) 1s^22p^4 (e) None of these | Homework.Study.com