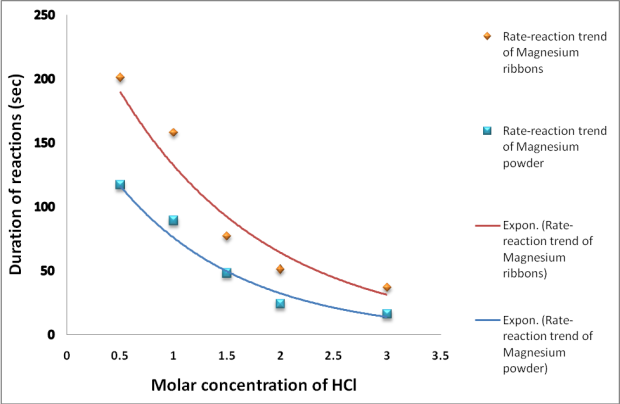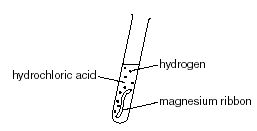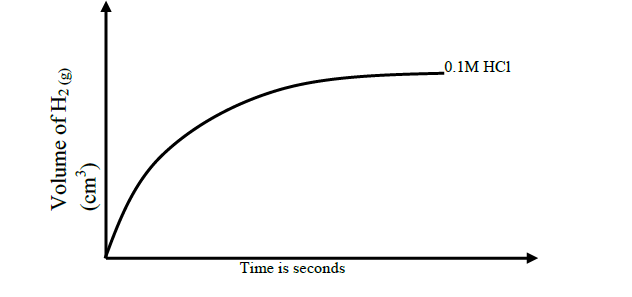
The sketch of the graph below shows a reaction between magnesium ribbon and dilute hydrochloric acid.
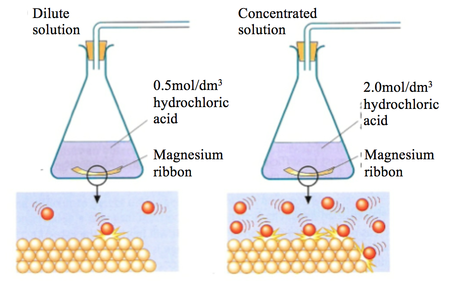
4.20 explain the effects of changes in concentration of solutions and pressure of gases on the rate of a reaction in terms of particle collision theory - iGCSE CHEMISTRY REVISION HELP

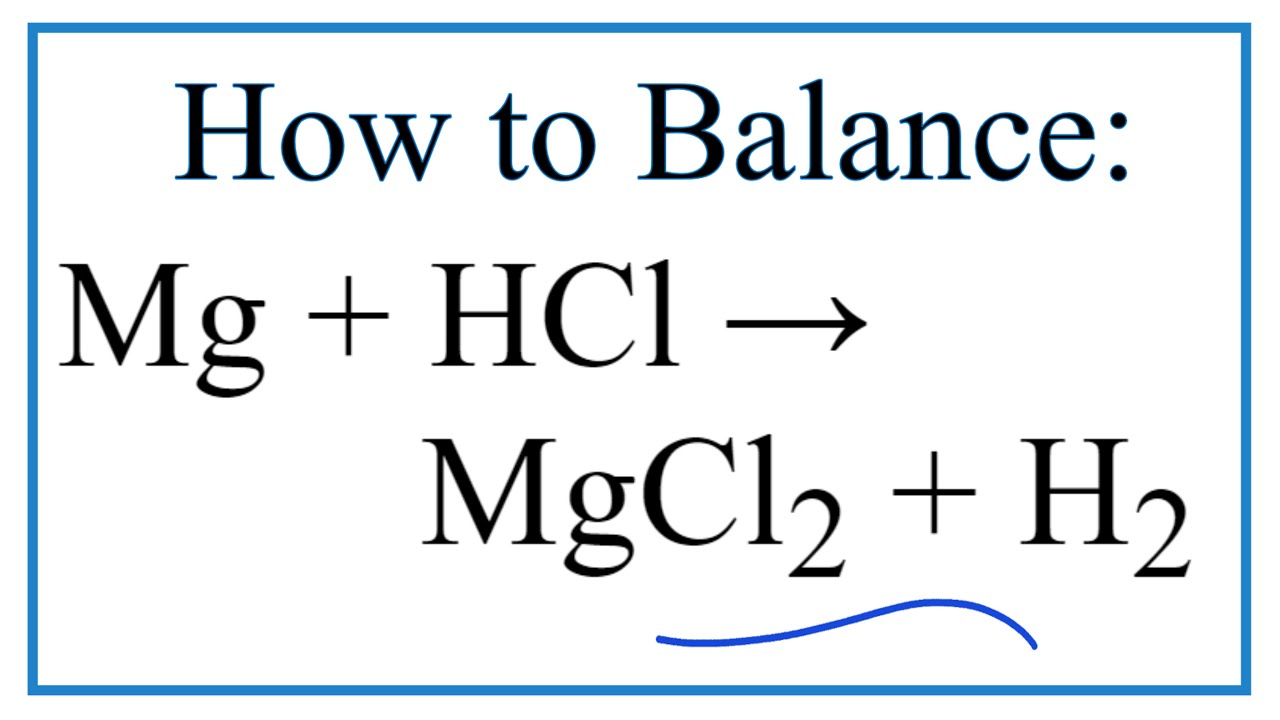
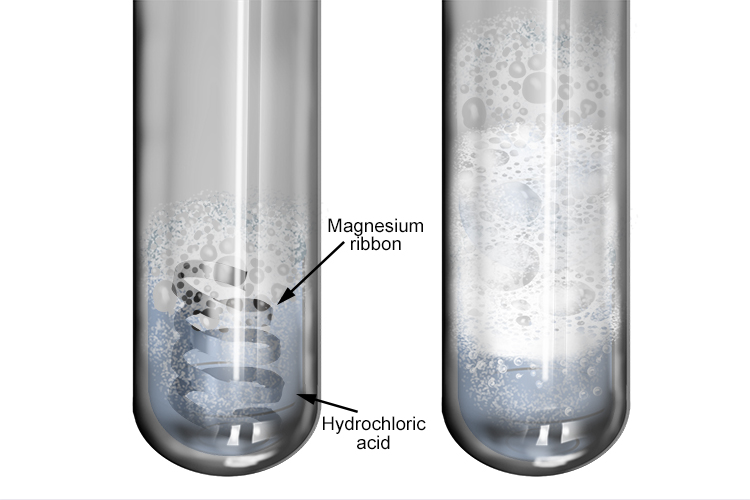
Bildagentur | mauritius images | Magnesium (Mg) ribbon in a beaker reacting with hydrochloric acid (HCl). This reaction creates magnesium chloride (MgCl2) and hydrogen gas (H2). Mg + 2HCl --> MgCl2 + H2