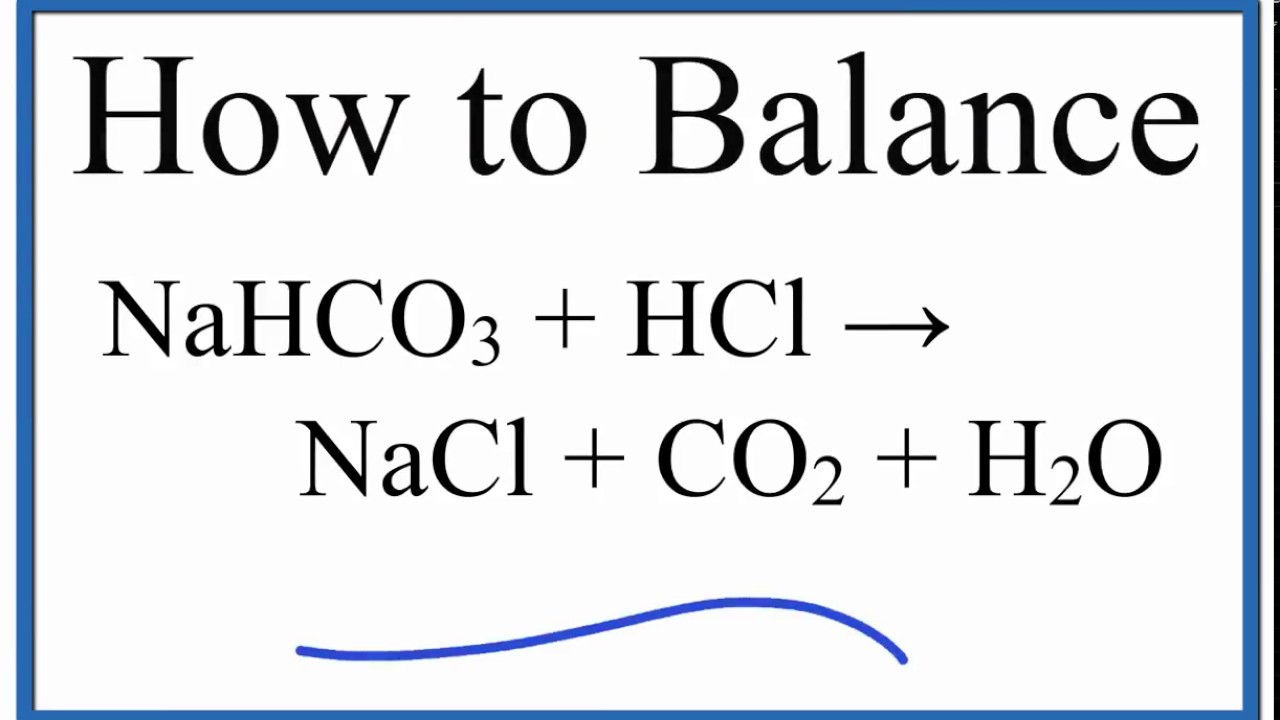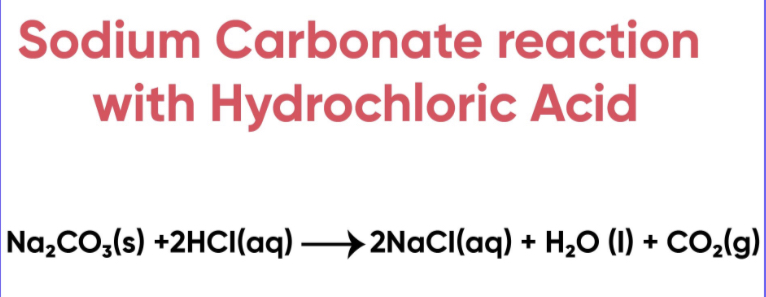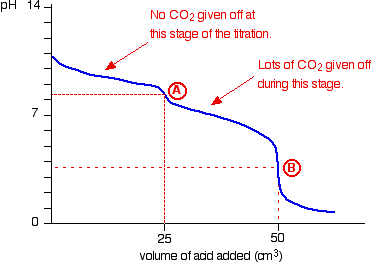
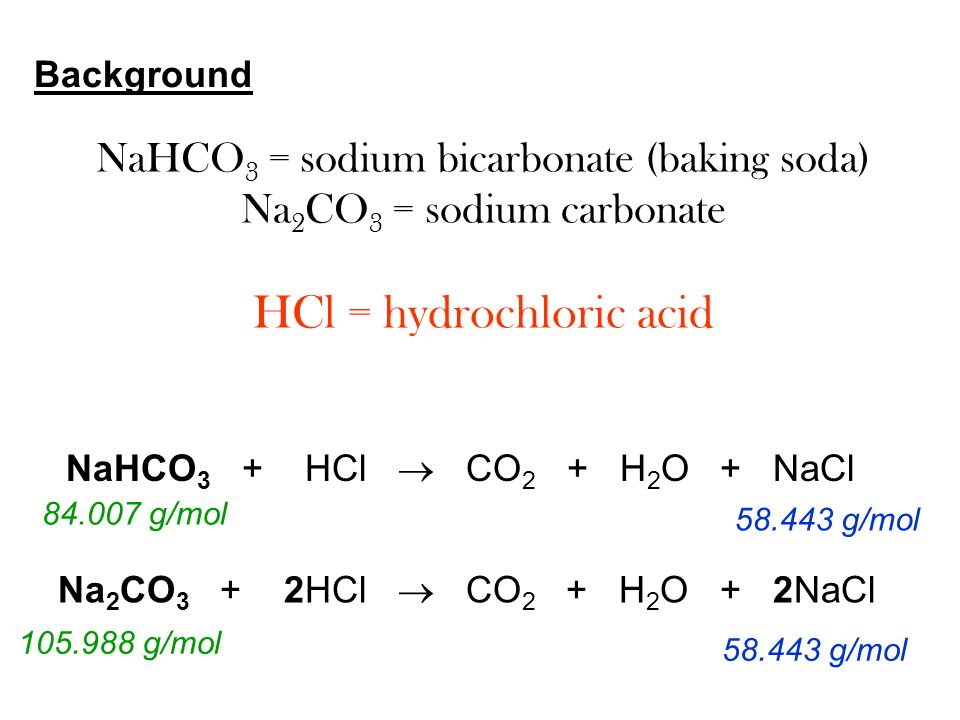
20.00 ml of 0.10 M sodium carbonate is placed in a flask with phenolphthalein and methyl orange indicators present. 0.10 M hydrochloric acid is placed in the burette and run in slowly.
Lab 8 Sodium Carbonate or Sodium Bicarbonate? Objective To determine a compound to be either Na 2 CO 3 or NaHCO ppt download

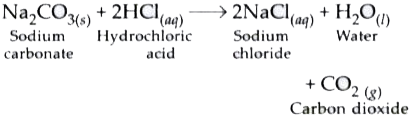
Explain in detail the reaction between sodium carbonate with hydrochloride acid with figure - Brainly.in

Determine the Heat of Reaction for a weak acid and carbonate salt. Sodium carbonate reacts with ethanoic acid according to the equation below: Na2CO3(s)+2CH3COOH(aq) | Homework.Study.com