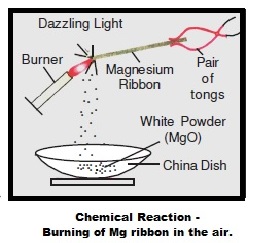
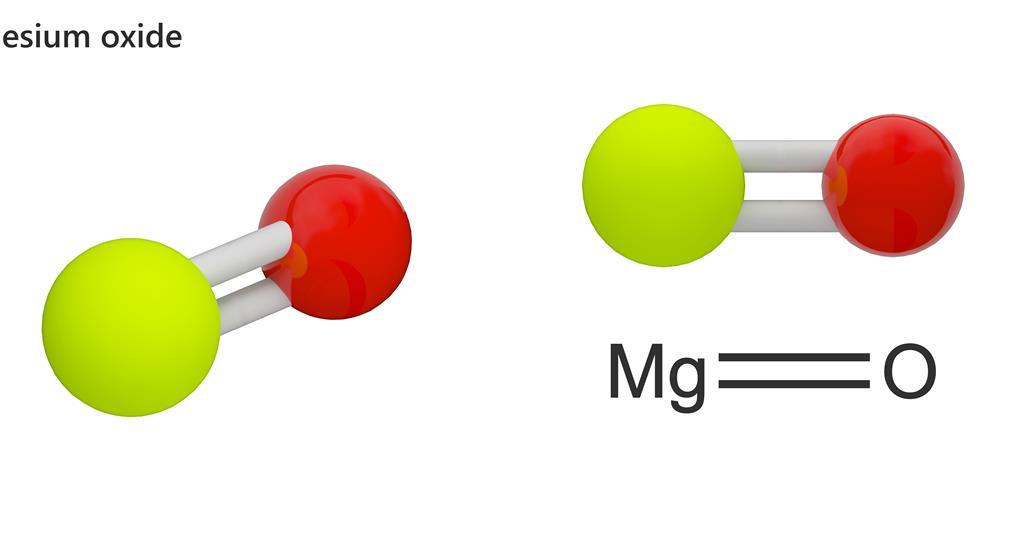A student performs an experiment of burning magnesium ribbon in the air. A chemical reaction takes place and as a result, a white powder X forms along with a bright white light.The

Burning of magnesium ribbon in air and collection of magnesium oxide in a watch glass experiment - YouTube

Chemical Equations. When chemicals are added together and a reaction occurs a chemical equation can be written The reactants go on the left of the equation. - ppt download

Write a balanced chemical equation for the following chemical reaction : Magnesium burns in oxygen - YouTube

A magnesium ribbon is burnt in oxygen to give a white compound X accompanied by emission of light. If the burning ribbon is now placed in an atmosphere of nitrogen, it continues

SOLVED: ( Magnesium ribbon Vinegar Reaction 9. Mg(s) HCHOr(aq) Mg(CHHOz)(aq) Hz(g) g. Cooled solid product of Reaction 8 + Water Reaction 10. CuSO (s) HO(l) > CuSOs SHO(s)

Q1.When magnesium ribbon burns in air or oxygen, a product is formed . State the type of chemical - Brainly.in

Write balanced and informative chemical equations for the following reactions. Magnesium ribbon burns in air to produce magnesium oxide.
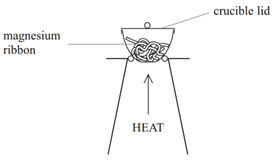
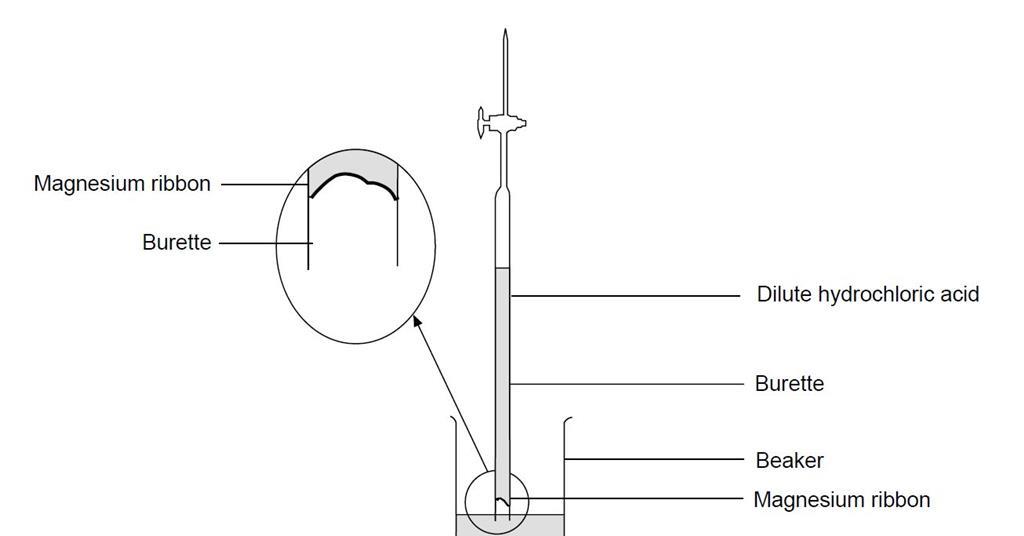
1:36 practical: know how to determine the formula of a metal oxide by combustion (e.g. magnesium oxide) or by reduction (e.g. copper(II) oxide) - TutorMyself Chemistry