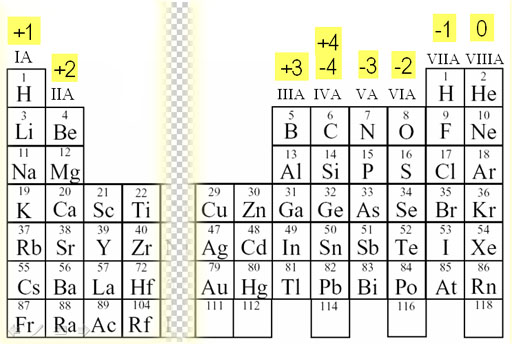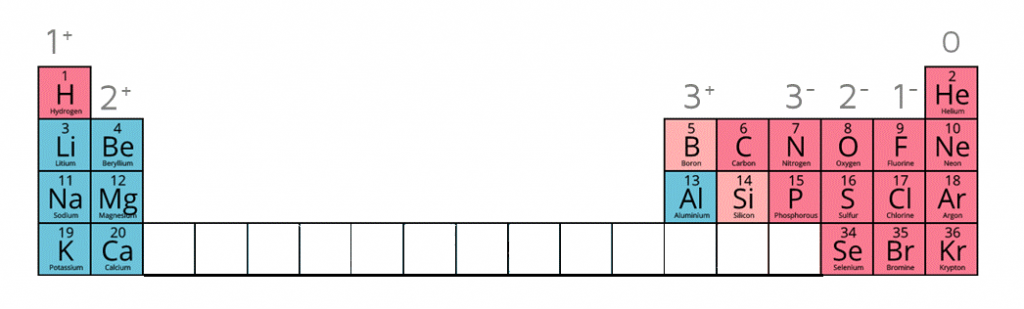
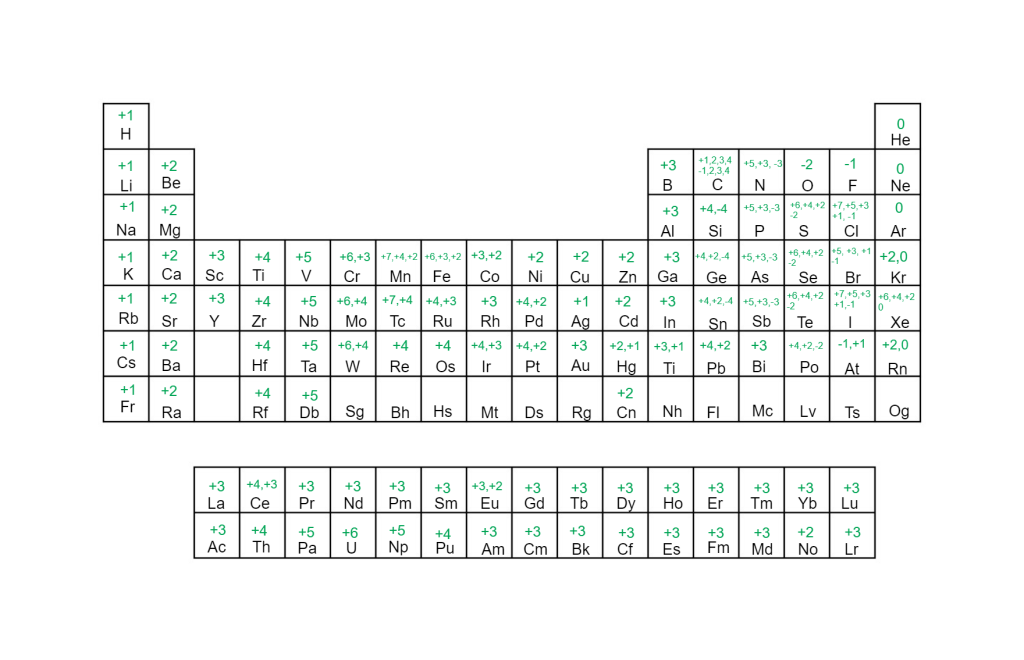
1:38 know the charges of these ions: metals in Groups 1, 2 and 3, non-metals in Groups 5, 6 and 7, Ag⁺, Cu²⁺, Fe²⁺, Fe³⁺, Pb²⁺, Zn²⁺, hydrogen (H⁺), hydroxide (OH⁻), ammonium (

aluminum periodic table charge | Periodic table of the elements, Periodic table, Periodic table poster
Working out the charges of ions - Using equations to represent chemical reactions - GCSE Chemistry (Single Science) Revision - OCR 21st Century - BBC Bitesize
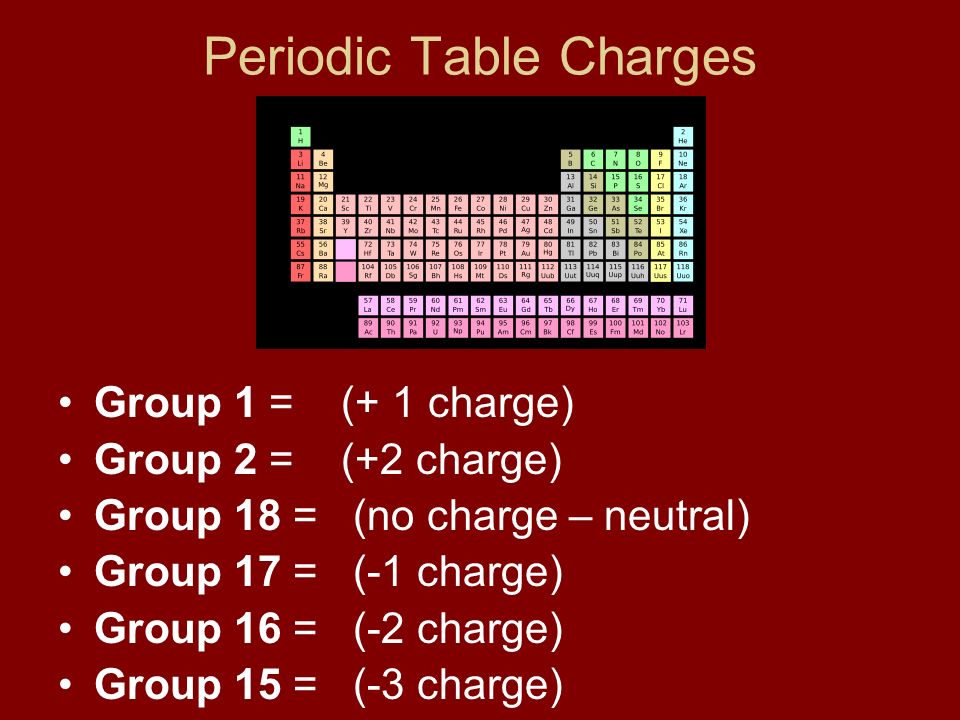
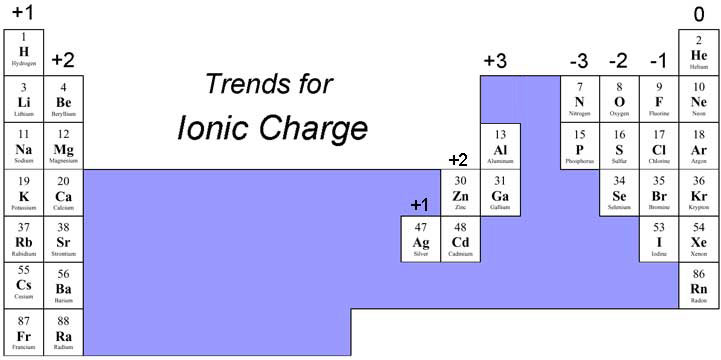
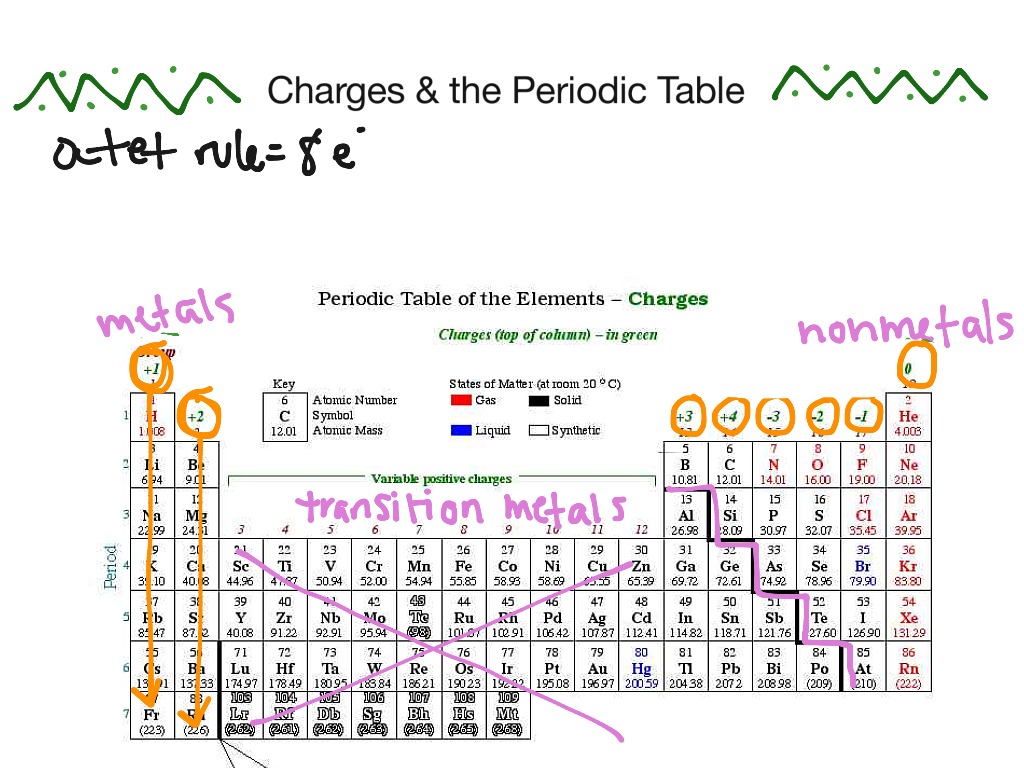
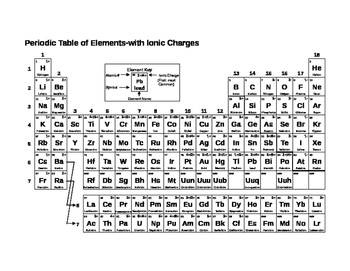

:max_bytes(150000):strip_icc()/PeriodicTableCharge-WBG-56a12db23df78cf772682c37.png)

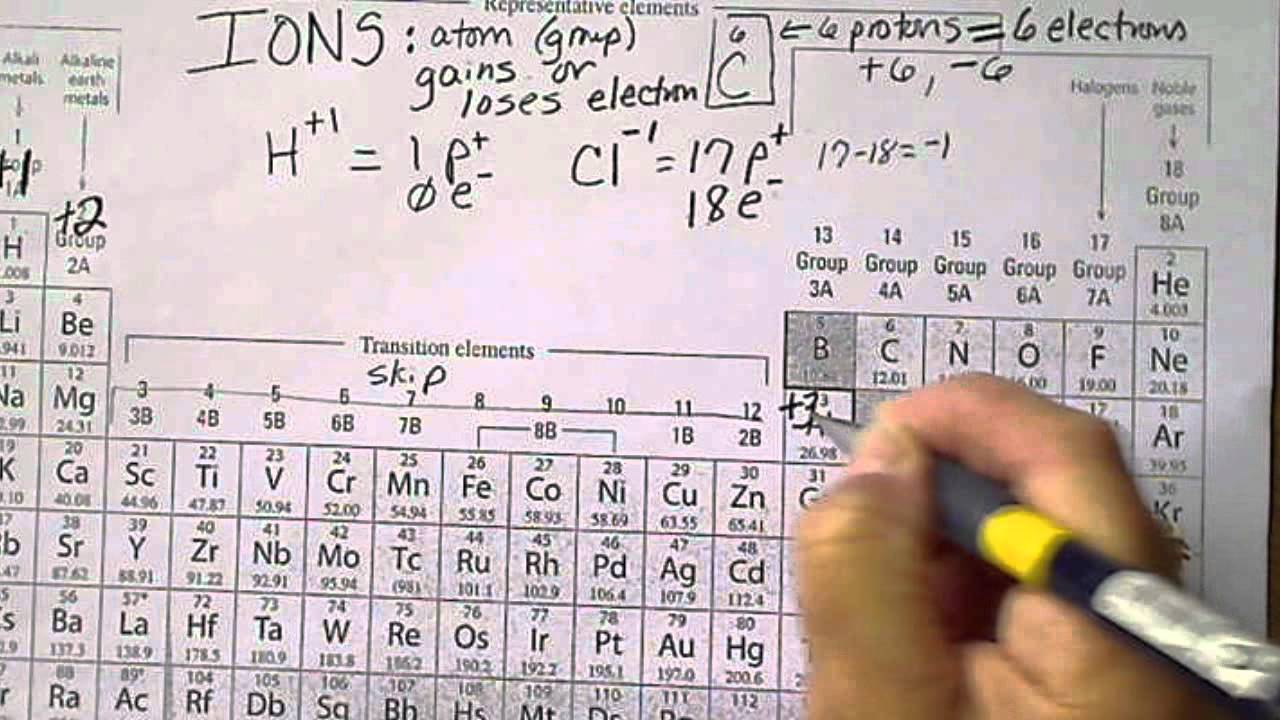





:max_bytes(150000):strip_icc()/GettyImages-186451118-589c07503df78c475854b41d.jpg)

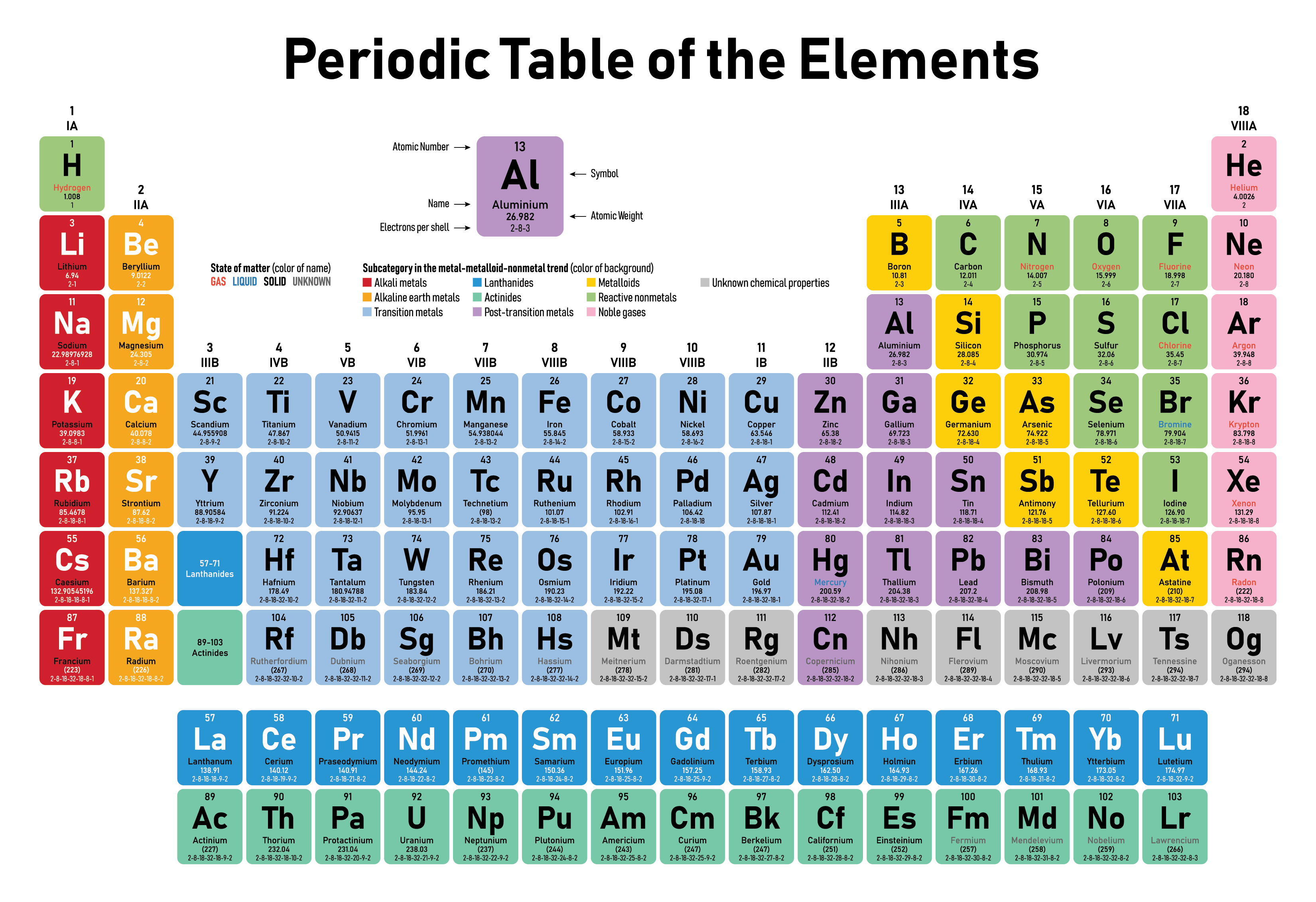
:max_bytes(150000):strip_icc()/PeriodicTableallcolor-58b5c82c3df78cdcd8bbb80f.png)