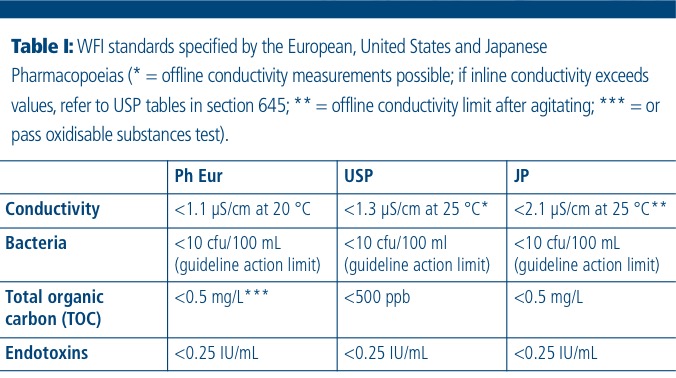
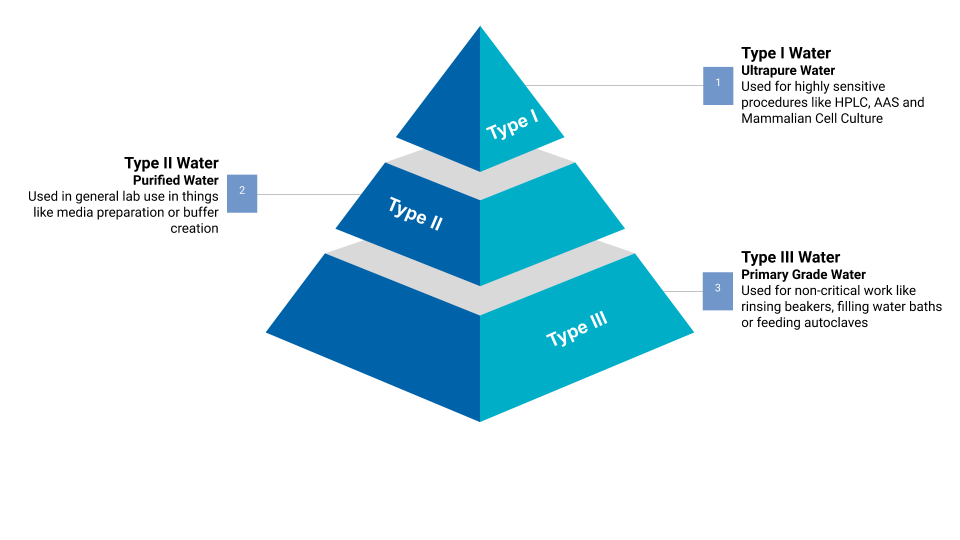
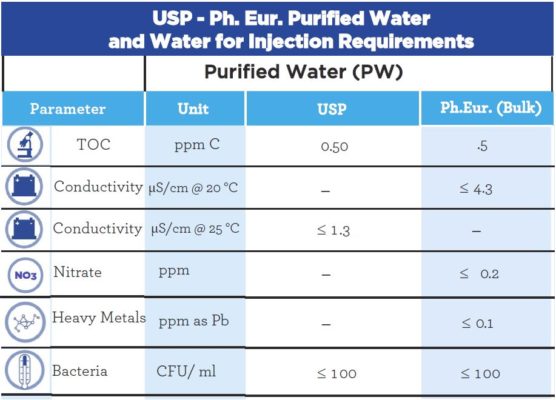
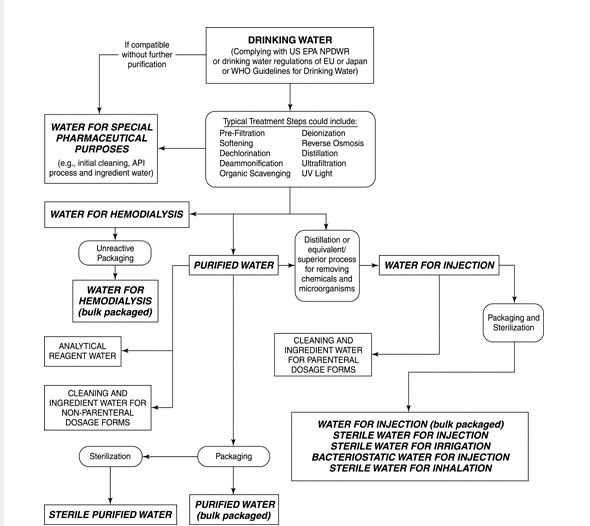
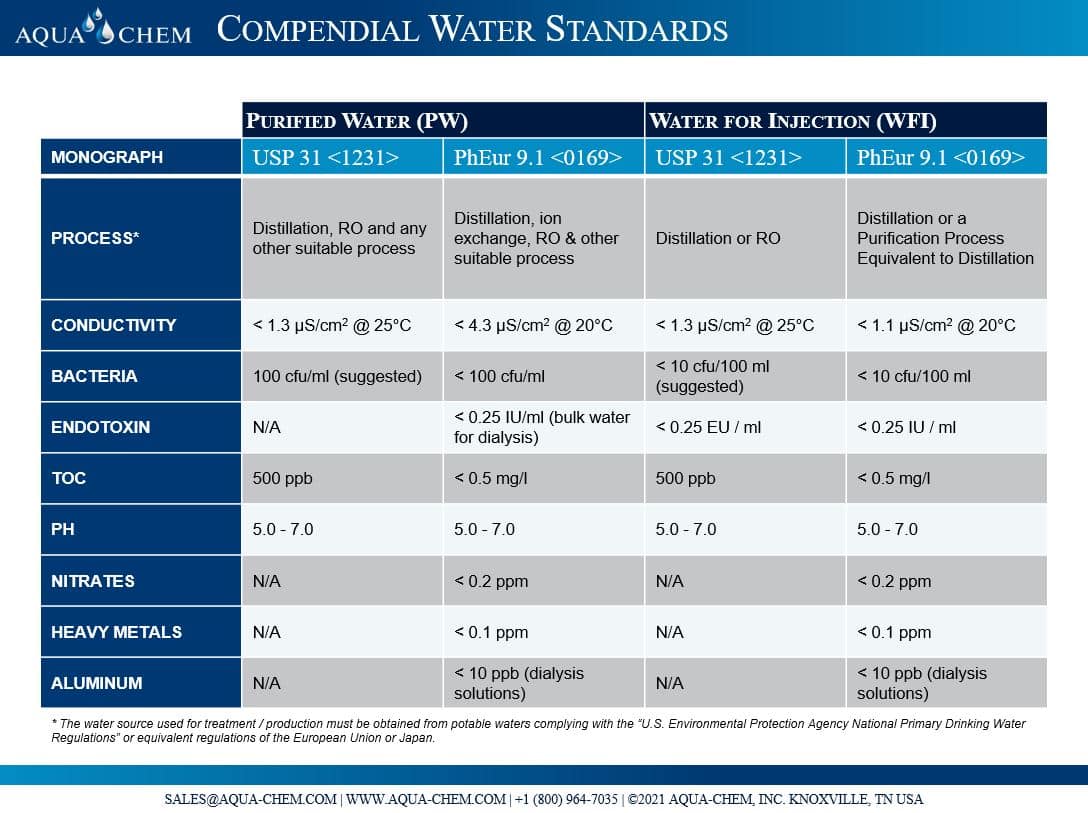
Occurrence and fate of endotoxin activity at drinking water purification plants and healthcare facilities in Japan - ScienceDirect

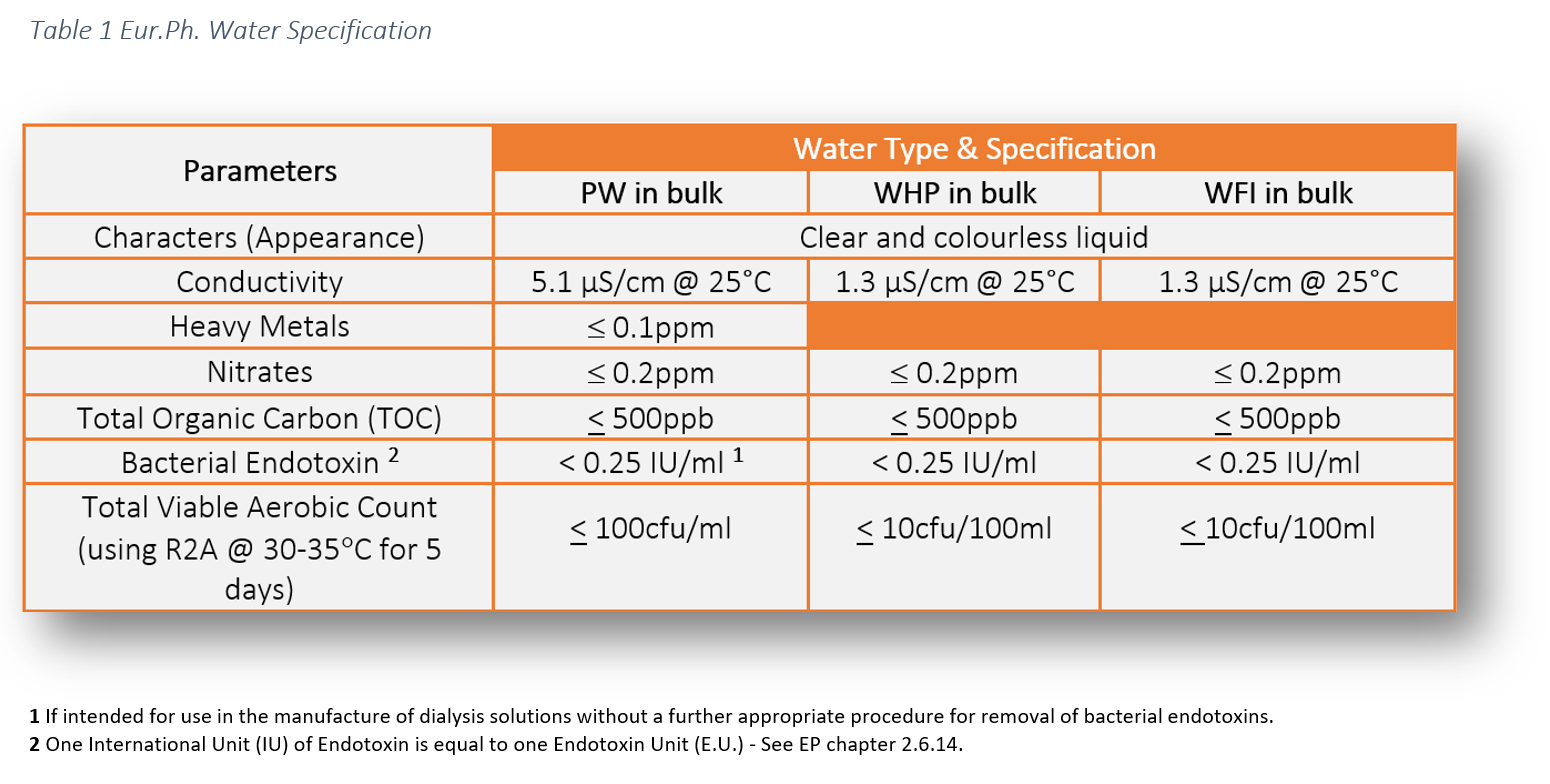
The Design, Control, Monitoring and Validation of Water Systems for Pharmaceuticals, Biologics, Medical Devices, Cosmetics, and Personal Care Products

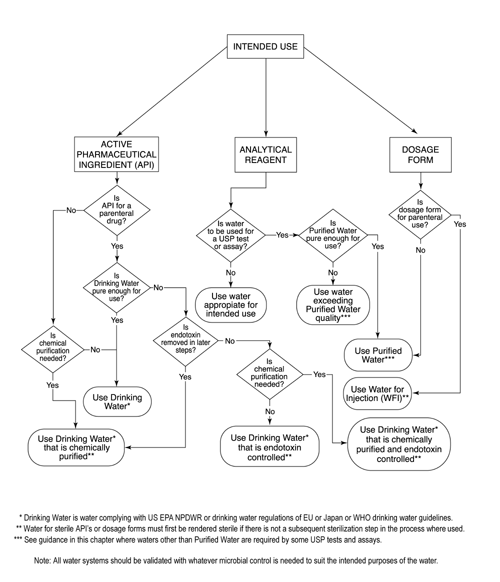
Current technologies to endotoxin detection and removal for biopharmaceutical purification - Schneier - 2020 - Biotechnology and Bioengineering - Wiley Online Library