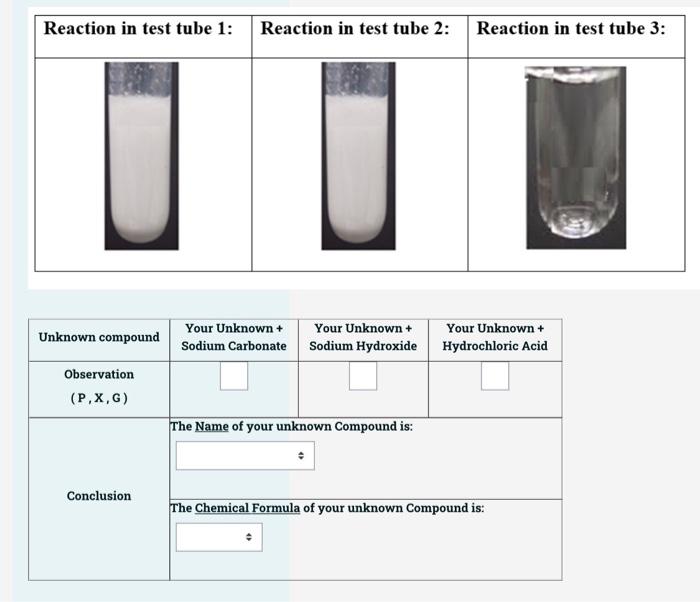
describe briefly the preparation and properties of sodium hydrogen carbonate. why is sodium hydrogen carbonate usedpowder in making baking ?

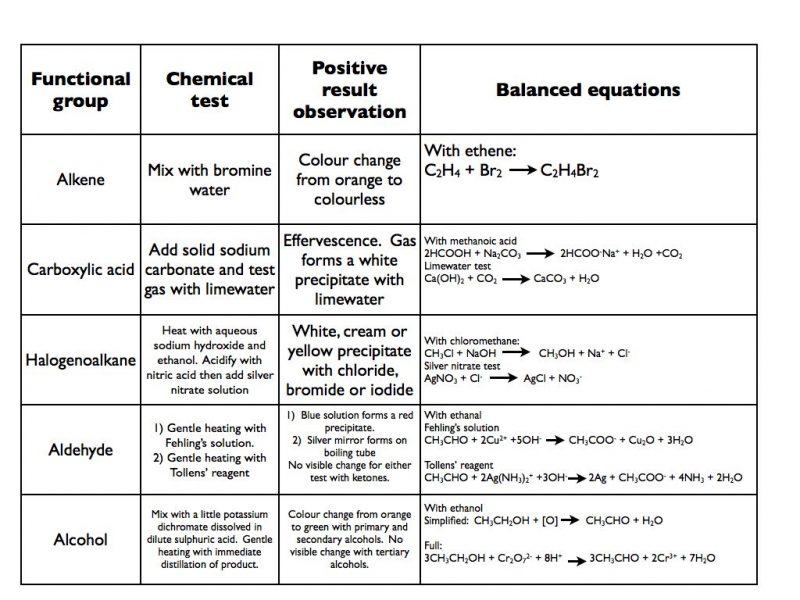
SOLVED: Na2CO3, Carboxylic acids react with sodium carbonate to form the sodium salt of the carboxylic acid, water, and carbon dioxide. Fizzing in the reaction can be viewed as a positive test

8) The reaction of acetic acid and sodium carbonate is shown below. Answer the questions with the of the diagram : Stand- Cork- Thistle funnel Gas delivery tube Small test tube O